 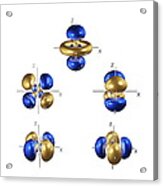 
A nodal surface is a plane or a sphere on which the electron can never be found the probability of the electron appearing on the nodal surface is zero. Note that although their shapes are different, they are roughly the same size, because they have the same energy.ģ) Most orbitals have at least one nodal surface. For example, here are the 90% contours of two orbitals that have the same energy. For example, here are the 90% contours of the simplest-looking orbitals for energy levels 1, 2 and 3 in a hydrogen atom, along with the diameters of the 90% contours.Ģ) Some orbitals have the same energy, but are different shapes. When we do this for hydrogen, we notice a few things right away….ġ) Orbitals that have different energies are always different sizes. We can only calculate wave functions for atoms with one electron, so we can only draw pictures of orbitals for one-electron atoms. To depict an orbital, then, we can draw a picture of the region in which the electron spends 90% of its time this is called the 90% contour. Electrons spend the vast majority of their time close to the nucleus. However, this is extremely unlikely the probability of the electron being that far away is around 10 -2,502,324,325. The electron in a hydrogen atom could be one foot away from the nucleus. Visualizing an orbital can be tricky, because electrons can be just about anywhere. In other words, “an orbital” means “a map of where the electron tends to spend its time.” This map is provided by the wave function (Ψ), so “orbital” and “wave function” mean the same thing (more or less). An orbital is a distribution for an electron. 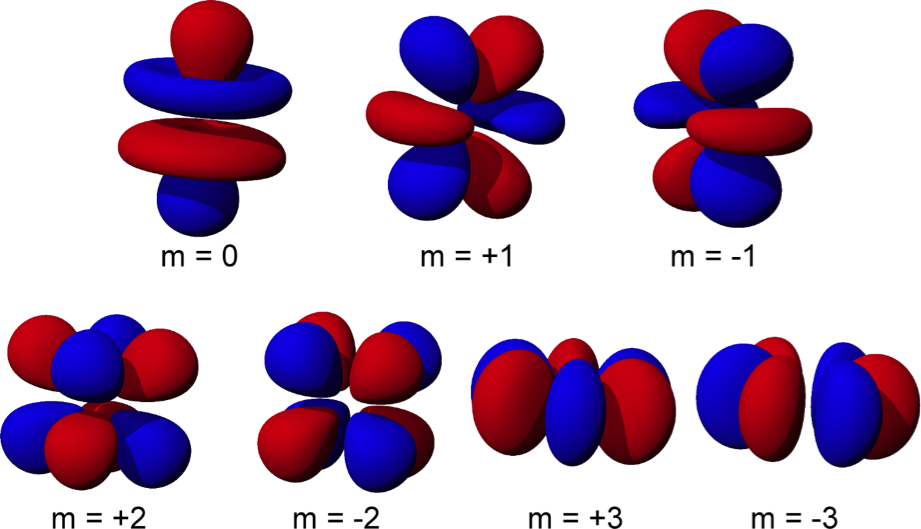
You’ve probably seen the term “orbital” in previous chemistry classes. Because the line never actually reaches the horizontal axis, the probability of finding the electron at very large values of r is very small but not zero. (b) In this plot of Ψ 2 versus r for the ground state of the hydrogen atom, the electron probability density is greatest at r = 0 (the nucleus) and falls off with increasing r. (a) The density of the dots shows electron probability. \) : Probability of Finding the Electron in the Ground State of the Hydrogen Atom at Different Points in Space. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
